1成果简介
准确提取电生理信号对于推进精神疾病发病机制的研究至关重要。然而,当前的生理电极仍面临诸多挑战,包括介电常数不匹配、生物相容性差以及信噪比不佳等问题。已有充分证据表明,孔隙结构调控是制备高性能生理电极的关键。本文,东华大学陈国印 副教授等在《ADVANCED FUNCTIONAL MATERIALS》期刊发表名为“Graphene Fiber-Based Bio-Microelectrode for Precise Neural Signal Recording”的论文,研究提采用非液晶纺丝技术结合剪切诱导取向工艺,制备了具有取向多孔结构的石墨烯纤维,其展现出高电导率(109.73 S cm?1)、适宜的界面阻抗(14.23Ω mm2)以及高电荷存储容量(82.89 mC cm?2)。在涂覆 SiO2和水凝胶层后,石墨烯纤维实现了侧面的电绝缘,防止了电荷泄漏,并建立了自适应界面,有效降低了界面模量。因此,它可作为生物微电极,用于精确检测目标脑区的神经活动,且提取的电生理信号具有 12.83 dB 的高信噪比。本研究提出了一种可扩展且协同的策略,用于构建下一代生物微电极,能够以相对较小的侵入性损伤高质量地提取电生理信号。
2图文导读
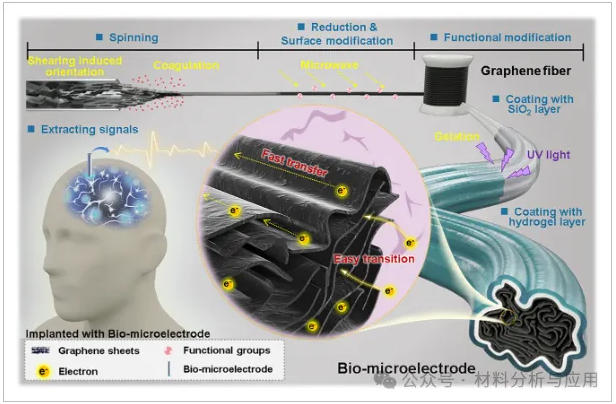
方案一、Design of a bio-microelectrode for physiological electrical signal acquisition.
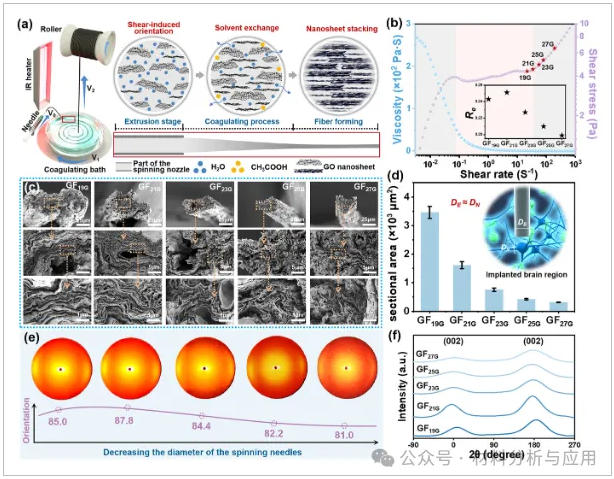
图1、Preparation and structural characterization of tunable-orientation porous graphene fibers: (a) Schematic illustration of the fabrication process and the oriented arrangement of graphene sheets; (b) Dynamic viscosity and shear stress variations of the spinning solution at varying shear rates, the inset shows the Reynolds number variation; (c) SEM images of the fabricated GFs, scale bars from top to bottom are 25, 5, and 1 μm, respectively (mean ± SD, n = 5); (d) Cross-sectional area of the fabricated GFs; (e) 2D X-ray diffraction patterns along the radial direction of GFs; (f) Normalized scattering intensity profiles.
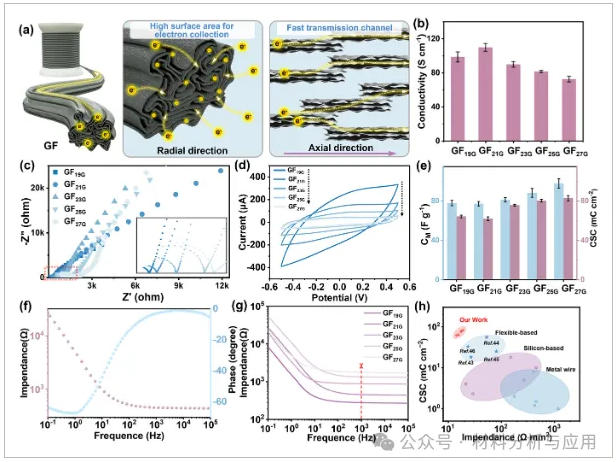
图2、Electrochemical performance regulation of GFs: (a) The influence of layer orientation on electron transport and the effect of porous structure on charge storage; (b) Conductivity of GFs measured by the four-electrode method (mean ± SD, n = 5); (c) Nyquist plots of all GFs obtained by EIS (x-axis: real part of impedance Z′, y-axis: imaginary part of impedance -Z″) (test length: 1.5 cm); (d) Cyclic voltammetry curves of GFs in the potential range of ?0.5 to 0.5 V, with a scan rate of 50 mV s?1; (e) Charge storage capacity of GFs (mean ± SD, n = 5); (f) Bode plot of GF21G; (g) Impedance spectroscopy of GFs (test length: 1.5 cm) at 1 kHz; (h) Comparison of specific impedance and charge storage capacity between GFs and other similar electrode materials.
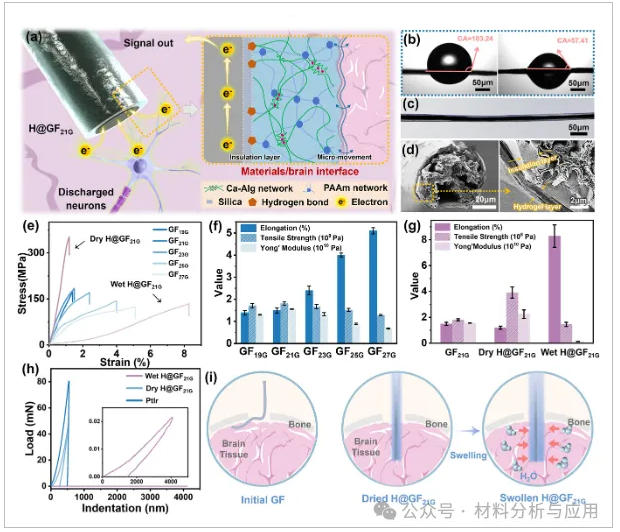
图3、Mechanical performance regulation of GFs: (a) Hydrogel and SiO2 coating for modulus matching and electrical insulation respectively; (b) Contact angle changes of GFs after plasma treatment (mean ± SD, n = 5); (c) Optical micrograph and (d) cross-sectional SEM image of H@GFs; (e) Stress–strain curves of GFs and H@GFs; (f, g) Elongation, tensile strength, and Young's modulus of (f) GFs and (g) H@GFs (mean ± SD, n = 5); (h) Mechanical properties of wet-H@GFs, dry-H@GFs, and PtIr microfibers measured by nanoindentation (mean ± SD, n = 3); (i) Implantation capability and modulus matching before and after coating.
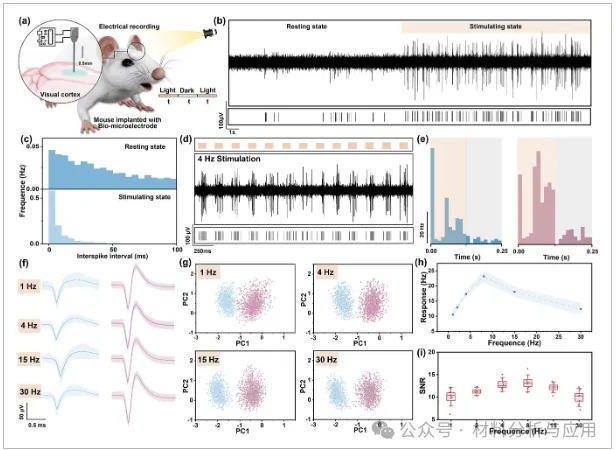
图4、In vivo evaluation of visual cortex signal recording: (a) Schematic showing the implantation of H@GF21G into the specified region of the mouse visual cortex; (b) Band-pass filtered visual evoked response waveforms and (c) interspike interval distribution of the recorded neurons under both dark resting conditions and light stimulation; (d) Real-time recording of neural activity under 4 Hz light stimulation displayed from top to bottom: visual stimulus marker, band-pass filtered waveform (300 ~ 5000 Hz), and spike markers indicating action potentials evoked by light stimulation; (e) Post-stimulus time histogram for analyzing neuronal firing rates; (f) Waveforms of sorted single-unit spikes across different stimulation periods; (g) Principal component analysis of the spikes in (f); (h) Time-frequency tuning curves under varying stimulation frequencies; (i) SNR of units across stimulation frequencies.
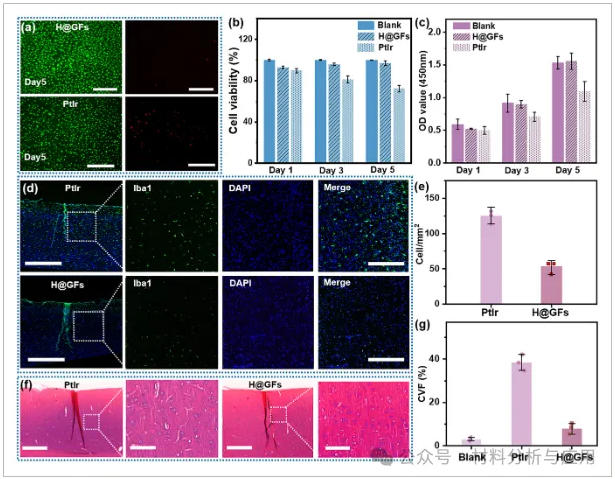
图5、Biocompatibility evaluation of H@GFs and PtIr: (a) Immunofluorescence live/dead staining of the Blank group, H@GFs group, and PtIr group on day 5, scale bar = 10 μm; (b) Cell viability; (c) OD value of the cytotoxicity test; (d) Immunofluorescence images of brain tissue 30 days post-electrode implantation. PtIr microelectrodes (top) and H@GF21G microelectrodes (bottom) were implanted in the mouse brain. Blue: DAPI (cell nuclei); Green: Iba1 (microglia). Three independent animals were included in each test group; (e) Quantitative analysis of inflammatory cell density surrounding the implanted electrode; (f) Masson's trichrome staining image depicting the implantation site 30 days post-implantation, including the implantation site (scale bar = 100 μm) and close-up images (scale bar = 20 μm); (g) CVF calculated from (f).
3小结
总之,本研究成功开发了一种弹性模量可调的微纤维神经电极。通过调节石墨烯片层的孔径和取向,我们制备出了尺寸与单个神经元相当的石墨烯纤维。这些纤维具有高导电性、低比阻抗和优异的电荷存储能力。此外,通过表面改性并涂覆稳定的双网络水凝胶,该电极在干燥状态下展现出类金属的力学性能,便于植入;而在湿润状态下,其弹性模量与脑组织相匹配。这种设计有效防止了电荷泄漏,确保了生物力学界面的稳定性,并展现出优异的生物相容性。基于这些结构创新和性能优化,该电极在小鼠视觉皮层中可靠地记录了单个神经元的动作电位信号,并在不同频率的光刺激下实现了高时间分辨率。它为柔性神经电极及设备-组织接口的设计提供了系统性解决方案,在神经系统监测和精神疾病管理方面展现出重要价值。此外,通过先进的空间或多功能集成,该柔性生理电极的结构可进一步优化,为复杂的神经生物学研究提供了广阔的前景。
文献
